This is the pre-print manuscript of an article published in the Kirk-Othmer Encyclopedia of Chemical Technology. The published version of the article is available at the Wiley website.
How to cite: Costanzi, S. (2020). Chemical Warfare Agents. In Kirk‐Othmer Encyclopedia of Chemical Technology, (Ed.). doi:10.1002/0471238961.0308051308011818.a01.pub3
A PDF version of the manuscript is available here.
Stefano Costanzi
Department of Chemistry and Center for Behavioral Neuroscience
American University, Washington, D.C.
Abstract. Chemical weapons are weapons that exploit the toxicity of chemicals to bring about death or harm. The toxic chemicals on which chemical weapons are based are known as chemical warfare agents. This article starts by providing some definitions of chemical weapons and chemical warfare agents as well an overview of key toxicology concepts and some notes on the physicochemical properties of the agents. Subsequently, it illustrates the chemistry, biochemistry, and medical countermeasures of the main classes of chemical warfare agents, namely vesicants, choking agents, blood agents, irritants, nerve agents, and centrally incapacitating agents.
Chemical weapons are weapons that exploit the toxicity of chemicals to bring about death or harm. The toxic chemicals on which chemical weapons are based are known as chemical warfare agents. The elimination of this entire category of weapons is the aim of the Convention on the Prohibition of the Development, Production, Stockpiling and Use of Chemical Weapons and on their Destruction, also known as Chemical Weapons Convention or CWC, which was opened for signature in 1993 and entered into force in 1997. Administered and implemented by the Hague-based Organisation for the Prohibition of Chemical Weapons (OPCW), the CWC is an international treaty that enjoys almost universal embracement, having been ratified or acceded by 193 States Parties. Importantly, the CWC poses a complete and absolute ban on chemical weapons, mandating State Parties to renounce “(a) to develop, produce, otherwise acquire, stockpile or retain chemical weapons, or transfer, directly or indirectly, chemical weapons to anyone; (b) to use chemical weapons; (c) to engage in any military preparations to use chemical weapons; (d) to assist, encourage or induce, in any way, anyone to engage in any activity prohibited to a State Party” under the Convention (CWC Article II, Paragraph 1) (1-3). This complete ban marks a substantial departure from previous international treaties or national policies on chemical weapons, which prohibited exclusively their use, chiefly intended, either implicitly or explicitly, as “first use” of these weapons – for instance, see the 1899 Hague Gas Declaration, the 1925 Geneva Protocol, or Richard Nixon’s 1969 “Statement on Chemical and Biological Defense Policies and Programs” (4-6). The complete ban posed on chemical weapons by the CWC mirrors an analogous ban posed on biological weapons and toxins by the Biological Weapons Convention (BWC), which was opened for signature in 1972 and entered into force in 1975. In fact, the BWC explicitly mandated State Parties to conduct negotiations in good faith to reach a similar agreement on the prohibition of chemical weapons (BWC Articles IX and XII) (7).
Despite the complete ban posed by the CWC, chemical weapons are far from being relegated to the past. After the entrance into force of the CWC in 1997, for a period of about 15 years, there were no reports of chemical weapons usage. However, beginning in 2012, chemical weapons have been used in several occasions by state and non-state actors as weapons of mass destruction or for targeted assassinations. In particular, they have been used in the Syrian civil war by both state and non-state actors (including the notorious Ghouta attack in August 2013, attributed to the Syrian regime by the United Nations, which led to the death of about 1400 people), in Iraq by non-state actors, in Malaysia for the assassination of Kim Jong-nam, half-brother of North Korea’s leader Kim Jong-un, in February 2017, and in the United Kingdom for the attempted assassination of Sergei Skripal and his daughter in March 2018. This recent trend in the use of chemical weapons has led international security and arms control experts to think about ways of restoring the restraint on the use of chemical weapons. At the same time, the international community formed the International Partnership against Impunity for the Use of Chemical Weapons, launched in Paris in January 2018, with the aim of supplementing the existing international mechanism to combat the proliferation of chemical weapons (6,8-11).
This article starts by providing some definitions of chemical weapons and chemical warfare agents as well an overview of key toxicology concepts and some notes on the physicochemical properties of the agents. Subsequently, it illustrates the chemistry, biochemistry, and medical countermeasures of the main classes of chemical warfare agents, namely vesicants, choking agents, blood agents, irritants, nerve agents, and centrally incapacitating agents.
This article does not cover personal protective equipment, detection, chemical and biochemical analyses, or decontamination. Similarly, it does not cover biological toxins, beyond discussing their coverage by the CWC in Section 1.1. For these matters, the reader is referred to other sources (12-20).
1. Definitions
1.1. Chemical weapons and chemical warfare agents. Chemical weapons are weapons intended to cause death or incapacitation through the toxic properties of chemicals. Chemical weapons are clearly defined in Article II of the CWC, according to which chemical weapons “means the following, together or separately: (a) toxic chemicals and their precursors, except where intended for purposes not prohibited under this Convention, as long as the types and quantities are consistent with such purposes; (b) munitions and devices, specifically designed to cause death or other harm through the toxic properties of those toxic chemicals specified in subparagraph (a), which would be released as a result of the employment of such munitions and devices; (c) any equipment specifically designed for use directly in connection with the employment of munitions and devices specified in subparagraph (b)” (CWC Article II, Paragraph 1) (1,3).
Of extreme importance to understanding chemical warfare agents which, as explained are the toxic chemicals on which chemical weapons are based, is the second paragraph of Article II of the CWC. According to this paragraph, toxic chemicals are defined as “any chemical (emphasis added) which through its chemical action on life processes can cause death, temporary incapacitation or permanent harm to humans or animals. This includes all such chemicals, regardless of their origin or of their method of production, and regardless of whether they are produced in facilities, in munitions or elsewhere…” (CWC Article II, Paragraph 2) (1).
The definition of chemical weapons provided by the CWC is broad and comprehensive. Any weapon designed to bring about death, temporary incapacitation, or permanent harm through the toxic properties of chemicals is considered a chemical weapon. As one can notice, the CWC does not establish any toxicity threshold in its definition of toxic chemicals. In fact, toxicity thresholds cannot be established because the toxicity of chemicals is entirely dependent on the amount of the chemical (the dose) to which an individual is exposed. As it had already been noted by Paracelsus, “all things are poison, and nothing is without poison; only the dose permits something not to be poisonous” (21). In other words, if administered at a high enough dose, all substances can exert a toxic effect. This implies that comprehensive lists of chemical warfare agents cannot be established. Some chemicals, for instance nerve agents, have no known legitimate uses. Hence, it is straightforward to classify these chemicals as tout court chemical warfare agents. Other chemicals, for instance chlorine or phosgene, have many legitimate uses in chemical industry. Despite their legitimate uses, such dual use chemicals are indeed considered chemical warfare agents if they are employed to intentionally cause death, incapacitation, or harm through their toxicity, and weapons based on such chemicals are indeed considered chemical weapons. Beyond the toxic chemicals themselves, paragraph 1 of Article 2 of the CWC extends the definition of chemical weapons to their precursors, i.e. the chemicals used to synthesize them (1). Some precursor chemicals have little or no use beyond serving as reactants for the synthesis of chemical warfare agents. However, in the majority of cases, precursors are dual use chemicals that, beyond their role in the synthesis of chemical warfare agents, also have legitimate, peaceful applications. Just as in the case of dual use toxic chemicals, dual use precursors indeed fall within the CWC prohibitions if they are intended to be employed for the synthesis of chemical warfare agents.
In its Annex on Chemicals, the CWC lists three schedules of chemicals, Schedule 1, Schedule 2, and Schedule 3 (2). These schedules are not intended to provide a comprehensive list of chemical warfare agents and their precursors – as mentioned, that would be an unfeasible task, as any chemical becomes a chemical warfare agent when it is intentionally employed to kill or harm through its toxic properties. Instead, the schedules are intended to support the CWC verification regime, by identifying chemicals of concern that are subject verification measures under the auspices of the OPCW and must be declared by States Parties. Schedule 1 identifies chemicals that are considered exclusively or primarily as chemical warfare agents or chemical warfare agent precursors. Schedule 2 and Schedule 3 identify dual use chemicals of concern, i.e. chemical warfare agents and precursors that have also legitimate uses, for instance in the pharmaceutical or chemical industry. In particular, Schedule 2 and Schedule 3 identify toxic chemicals and precursors of concern that are produced for legitimate purposes not prohibited by the CWC in smaller quantities and large commercial quantities, respectively (22). As specified in the Verification Annex of the CWC, for Schedule 1 chemicals, States Parties are bound not to acquire, produce, retain, or use the listed chemicals unless for research, medical, pharmaceutical, or protective purposes, and in modalities and quantities that are consistent with such purposes, with the aggregate amount of such chemicals not exceeding 1 metric ton at any given time (23). For Schedule 2 chemicals, declaration by States Parties are required when the amount of chemicals produced, processed, or consumed in a given year exceeds thresholds set to 1 kg for toxic chemicals listed in Schedule 2, part A and marked with a star, 100 kg for all other toxic chemicals listed in Schedule 2, part A, and 1 metric ton for chemical precursors listed in Schedule 2, part B. Thresholds beyond which verification shall be carried out are set at 10 kg for toxic chemicals listed in Schedule 2, part A and marked with a star, 1 metric ton for all other toxic chemicals listed in Schedule 2, part A, and 10 metric tons for precursor chemicals listed in Schedule 2, part B (24). For Schedule 3 chemicals, declaration by States Parties are required when the yearly production of a given chemical exceeds a threshold of 30 metric tons, while verification shall be carried out when the aggregate production of any Schedule 3 chemical above the declaration threshold exceeds 200 metric tons (25).
By no means are chemical warfare agents limited to synthetic toxic chemicals prepared in laboratories, chemical plants, or other facilities. As clearly stated by the CWC, their origin or method of production does not affect the definition of toxic chemicals. As a result, biological toxins, i.e. toxic chemicals produced by living organisms, fit the definition of chemical warfare agents if they are used to bring about death, incapacitation of harm. Schedule 1 of the CWC explicitly lists two biological toxins, namely saxitoxin and ricin. Beyond these two toxins, just like for all other toxic chemicals, any other biological toxin intentionally used to cause death, incapacitation, or harm is also to be considered a chemical warfare agent. It is worth pointing out that, because they are produced by living organisms, biological toxins fall also under the scope of the BWC, which, as mentioned, poses an entire ban on biological weapons and toxins – in fact, the full name of the BWC is Convention on the Prohibition of the Development, Production and Stockpiling of Bacteriological (Biological) and Toxin Weapons and on their Destruction (7).
Another consequence of the fact that the origin of toxic chemicals is irrelevant to the CWC definition of chemical weapons is that toxic chemicals produced in munitions (or any other delivery system) at the time when the munitions is delivered, or mixed immediately prior to their use, are to be considered chemical warfare agents and such munitions, known as binary munitions, are to be considered chemical weapons (26). Binary munitions are based on chemical precursors endowed with relatively low toxicity, which, after reacting together, give rise to toxic chemical warfare agents. In the crudest designs, a binary munition is filled with freshly mixed precursors immediately prior to its the deployment (sometimes, one precursor is preloaded into the munition, while the second one is added before the firing of the munition). In the most advanced designs, the two precursors are preloaded in separate chambers within the binary munition, and automatically come into contact and react together after the firing of the munition (10,27,28). Beyond the munitions, the chemical precursors on which binary munitions are based also fall within the CWC ban, if they are developed, produced, or stockpiled with the intention to be employed as components of binary chemical weapons.
Just as it does not pose any limitation on the nature of chemical warfare agents, the CWC does not pose any limitation on the nature of delivery systems or equipment designed to be used in connection with chemical weapons. Therefore, chemical weapons are not limited to munitions containing chemical warfare agents. Conversely, any device intended to intentionally deliver toxic chemicals to cause death, incapacitation or harm is considered a chemical weapon. Among others, these would include gas cylinders, sprayers, or even crude devices such as the plastic bags filled with sarin that, in the notorious 1995 Tokyo subway attack, the Japanese cult Aum Shinrikyo punctured with umbrellas in five different train cars, letting the nerve agent evaporate within the confined space of the cars and leading to the death of 12 passengers and the injury of nearly 1000 (29). This lack of specificity in the definition of chemical weapons by the CWC is very important to avoid potential loopholes. For instance, in April 1915, during the Second Battle of Ypres, the Germans delivered chlorine gas toward the enemy trenches by opening over 5000 chlorine cylinders and letting the gas be carried westward by the wind. The Hague Convention, the treaty that at the time banned the use of chemical weapons in warfare, required the Contracting Powers “to abstain from the use of projectiles the sole object of which is the diffusion of asphyxiating or deleterious gases.” Since it did not involve the use of projectiles, the use of chlorine in the Second Battle of Ypres, technically, did not violate the Hague Convention (10,30).
1.2 Riot control agents. The CWC defines riot control agents as “any chemical not listed in a Schedule, which can produce rapidly in humans sensory irritation or disabling physical effects which disappear within a short time following termination of exposure” (CWC Article II, paragraph 7). The use of riot control agents for warfare purposes is not permitted by the CWC. In fact, the Convention states that a State Party has the right to request to receive assistance and protection if it considers that, inter alias, riot control agents have been used against it as a method of warfare (CWC Article X, paragraph 8, subparagraph b). However, the Convention explicitly mentions “law enforcement including domestic riot control purposes” among the purposes not prohibited (CWC Article II, paragraph 9, subparagraph d). Furthermore, the CWC mandates State Parties to declare to the OPCW any chemicals that they hold for riot control purposes, specifying “chemical name, structural formula and Chemical Abstracts Service (CAS) registry number, if assigned” (CWC Article III, paragraph 1, subparagraph e) (1).
From the CWC articles described above, it is clear that the use of riot control agents for law enforcement purposes is permitted by the CWC, while their use in warfare is not. But what chemicals can be employed as riot control agents? Based on the definition of riot control agents provided by the CWC, the Scientific Advisory Board (SAB) of the OPCW advised that exclusively sensory irritants with short-lived effects should be considered for use as riot control agents. Moreover, the SAB identified a list of 17 chemicals previously declared as riot control agents by States Parties that were deemed to meet the CWC definition, as well as 43 additional chemicals that were deemed not meet the definition, as they cause persistent harm or fall within the scope of one of the CWC Schedules. Finally, the SAB advised that longer-lasting irritants, as well as incapacitating agents that act on the central nervous system, should not be considered as fitting the definition of riot control agents (31). For more information on chemosensory irritants and incapacitating agents, see Sections 4.3 and 4.6 below.
1.3 Herbicides. As discussed, the CWC bans all weapons that are intended to cause death, temporary incapacitation, or permanent harm to humans or animals through the action of toxic chemicals on life processes. As a result, there are several weapons that, although based on chemicals, do not fall within the scope of the CWC. Among others, the reference to humans and animals in the second paragraph of Article II of the CWC, excludes herbicides. Herbicides are indeed toxic chemicals, but they are intended to be toxic for plants, not humans or animals. Nonetheless, in its preamble, the CWC does recognize “the prohibition, embodied in the pertinent agreements and relevant principles of international law, of the use of herbicides as a method of warfare” (1). Of particular relevance in this sense is the Environmental Modification Convention, which was opened for signature in 1977 and entered into force in 1978, and prohibits State Parties from engaging “in military or any other hostile use of environmental modification techniques having widespread, long-lasting or severe effects as the means of destruction, damage or injury to any other State Party” (32,33). To be sure, in the case that herbicides were to be intentionally directed against humans or animals to cause harm through their toxicity, they would definitely fall within the scope of the comprehensive CWC ban. This is a direct consequence of the broad definition of chemical weapons, which encompasses any toxic chemical intentionally used for such purpose, without any exclusion.
2. Brief notes on the quantification of toxicity.
Before reviewing the toxicity of specific chemical warfare agents in Section 4, it is useful to briefly introduce some fundamental toxicology concepts and illustrate some relevant ways in which toxicity is commonly quantified.
The toxicity of chemicals is due to a combination of their toxicodynamic properties, i.e. the relationship between the concentration of the toxic chemical at the site of action in the body and the subsequent biological response, and toxicokinetic properties, i.e. the relationship between the dose of the toxic chemical administered to an individual and the concentration of the toxic chemical at the site of action, which is determined by the chemical’s absorption, distribution, metabolism, and excretion (ADME properties). Toxicodynamics can be regarded as the study of how a toxic chemical affects the organism by altering its functioning, while toxicokinetics can be regarded as the study of how a chemical is affected by the organism. The toxicodynamic properties of a chemical are chiefly governed by molecular recognition, i.e. how the chemical interacts with macromolecular cellular targets, and chemical reactivity. The toxicokinetic properties are chiefly governed by the physicochemical properties of the chemical, which have a strong impact on its ADME properties, as well as molecular recognition, especially involving enzymes responsible for the metabolism of the chemical and transporters that move the chemical across biological membranes, and chemical reactivity. Together, toxicodynamics and toxicokinetics determine the ultimate toxic effect of the exposure of an individual to a toxic chemical (34,35).

Fig. 1. Quantal dose-response curves for the incapacitating (red curve) and lethal (green curve) effects of a toxic substance. Note the logarithmic scale of the x-axis, which leads to the sigmoidal shape of the dose-response curves (if the x-axis had a linear scale, the curves would have had a hyperbolic shape).
In toxicology and pharmacology, quantal dose-response curves are frequently used to evaluate the toxicity as well as the pharmacological potency of chemicals (Fig. 1). In a quantal dose response curve, for instance in a pharmacological study conducted on laboratory animals, subjects are administered a range of doses of the substance under examination, with the intent of registering what percentage of the exposed population experiences a given predefined effect (for instance blood pressure dropping below a certain level) at different doses of the substance. The dose of the substance is plotted on the x-axis, typically expressed in weight amount of the administered substance (e.g. in mg), over body weight of the subjects receiving the substance (e.g. in kg). The percentage of the exposed population in which the predefined outcome is observed is plotted on the y-axis. The potency of the substance is typically expressed in terms of ED50 (effective dose 50), i.e. the dose the causes the predefined effect (in our example blood pressure dropping below a certain level) in 50% of the population who received it. The lower the ED50, the higher the potency of the substance under study – less of the substance is needed to produce the predefined effect in 50% of the population. In toxicology, where the effects under study are toxic effects, the ED50 is typically called TD50 (toxic dose 50). Moreover, when studying the lethality of a substance, i.e. when the effect under study is the death of the subjects exposed to the substance, the potency is expressed in terms of LD50 (lethal dose 50), i.e. the dose the causes death in 50% of the population who received it. For chemical weapons, LD50 values are commonly used to express the potency of liquid chemical warfare agents, e.g. through percutaneous exposure. It is worth noting that, given that lethality studies cannot be done in humans, human LD50 values can only be roughly extrapolated from data obtained from animal models (36).
For gaseous or aerosol substances that are dispersed in the air and are absorbed through inhalation, rather than determining the dose received by the subjects, it is more straightforward to measure the concentration of the substance in the air and the duration of the exposure (37). The product of the concentration of the substance in the air and the duration of the exposure, will determine the dosage of the substance received by a given subject. Hence, quantal concentration response curves are commonly used, with the environmental concentration of the substance (e.g. in mg/m3) plotted on the x-axis and the percentage of subjects in which the expected outcome is observed plotted on the y-axis, with the caveat that all subjects have been exposed to the substance for the same amount of time (t). In this case, the potency of the substance is typically expressed in terms of EC50 (effective concentration 50) at a given exposure time, i.e. the concentration that causes the predefined effect (in our example blood pressure dropping below a certain level) in 50% of the population who were exposed to the substance for a given amount of time. In analogy with the ED50, the lower the EC50, the higher the potency of the substance under study. When studying the lethality of a substance, the potency is expressed in terms of LC50 (lethal concentration 50). For substances that follow Haber’s law, the product of the environmental concentration multiplied by the time of exposure that yields a given biological effect (for instance lethality in 50% of the exposed population) is constant: C • t = k (37). In other words, a substance that has an LC50 of 500 mg/m3for a 1-minute exposure (exposure to a concentration of 500 mg/m3 for 1 minute causes lethality in 50% of the population) will have an LC50 of 50 mg/m3for a 10-minute exposure (exposure to a concentration of 50 mg/m3 for 10 minutes causes lethality in 50% of the population). Thus, it is useful to introduce the concept of ECt50 and LCt50, respectively defined as the product of the effective or lethal concentration of the substance multiplied by the time of exposure. For substances that follow Haber’s law, the LCt50 is constant. In our example, a substance that has LC50 values of 500 mg/m3 for a 1-minute exposure and 50 mg/m3 for a 10-minute exposure will have an LCt50 of 500 (mg/m3) min. For chemical weapons, LCt50 values are commonly used to express the potency of gaseous or aerosol chemical warfare agents, e.g. through inhalation.
However, not all chemicals follow Haber’s law. Conversely, according to the ten Berge model, also known as toxic load model, the environmental concentration of a chemical must be raised to an exponent (n) for its product with the time of exposure to remain constant: Cn • t = k. The constant k is also known as the toxic load and the exponent n is also known as the toxic load exponent. The toxic load exponent depends on the chemicals as well as the route of administration. Chemicals with a toxic load exponent equal to 1 follow Haber’s law. For chemicals with a toxic load exponent greater than 1, the concentration in the air has a greater impact in determining the biological effect than the time of exposure, and the LCt50 increases as the exposure duration increases. In other words, exposure to higher concentrations for shorter periods of time has a higher toxic effect than exposure to lower concentrations for longer periods of time. Finally, for chemicals with a toxic load exponent less than 1, the time of exposure has a greater impact in determining the biological effect than the concentration of the chemical in the air, and the LCt50 decreases as the exposure duration increases. In other words, exposure to lower concentrations for longer periods of time has a higher toxic effect than exposure to higher concentrations for shorter periods of time. For many chemicals the toxic load exponent is greater than 1, chiefly due to the fact that the organism can effectively detoxify low doses of the chemical over the course of protracted exposure. Of note, the toxic load model, although more general than Haber’s law, is not universally applicable, as the toxic load exponent may vary under certain conditions, for instance very short exposure times or pulse exposure (37-39).
Just as the lethal potency of chemical warfare agents can be expressed in terms of LD50, the incapacitating potency of chemical warfare agents can be expressed in terms of ID50 (incapacitating dose 50), i.e. the dose the causes a given level of incapacitation in 50% of the individuals who received it. Similarly, the incapacitating potency of gases and aerosols can be expressed in terms of ICt50 (incapacitating concentration multiplied by exposure time 50). As we discussed, the toxic effect of chemicals is entirely dependent on the administered dose. Hence, incapacitating agents can indeed be lethal if administered at higher doses. What distinguishes incapacitating agents from lethal agents is the separation between their ID50 and their LD50 values (Fig. 1) – or the ICt50 and their LCt50 values, for vapors and aerosols. In particular, for incapacitating agents, the ID50 value must be significantly lower than the LD50 value, so as to minimize the risk of a lethal outcome when the incapacitating agents are deployed. This mirrors the situation for pharmaceuticals, where the ED50 of a drug, i.e. the effective concentration that causes the desired effect in 50% of the exposed population, must be significantly lower than its LD50 to allow safe administration of the drug at the therapeutically relevant doses. In other words, the therapeutic index of the drug, defined as the ratio of its LD50 divided by its ED50, must be as high as possible (31,36,40).
3. Physicochemical properties of chemical warfare agents.
The physicochemical properties of chemical warfare agents (Table 1) play a significant role in determining their characteristics. As mentioned, they have a substantial impact on the toxicokinetics of chemicals and, therefore, their toxic effects. While this is true for all chemical warfare agents, toxicokinetics, and therefore physicochemical properties, have a particularly strong influence on the toxic effects of those agents that tend to interact non-specifically with a multitude of cellular targets, for instance choking agents and vesicant (35,41).
The melting and boiling points of chemical warfare agents determine their physical state at the temperature and pressure under which they are employed, which, in turn, is a key determinant of their route of absorption as well as the suitable delivery systems. Gaseous chemical warfare agents are typically absorbed by inhalation. On the battlefield, liquid and solid chemical warfare agents are typically dispersed as aerosols and are absorbed through inhalation or contact with the skin or the eyes. Beyond the battlefield, on a smaller scale, e.g. when the agents are used for assassination purposes, relevant routes of exposure for liquid and solid chemical warfare agents include also ingestion and injection. The route of absorption has a significant impact on determining which organs and tissues will be affected the most by chemical warfare agents, especially, as mentioned, for chemicals that interact non-specifically with a multitude of targets, such as choking agents and vesicants. Choking agents, which are found in the gaseous state at Normal Temperature and Pressure (NTP), are primarily absorbed through inhalation. Hence, the damage caused by them is chiefly centered on cells and tissues of the airways. Conversely, blister agents, which are found in the liquid state at NTP and are typically dispersed as aerosols, are absorbed not only through inhalation, but also through contact with the skin and the eyes. Hence, blister agents broadly damage airways, skin, and eyes. For more information on choking agents and blister agents, see Sections 4.2 and 4.1 below (37,42).
Vapor density is another physicochemical property that significantly affects the characteristics of chemical warfare agents. In particular, agents that have a vapor density higher than the density of air, e.g. chlorine, stay low to the ground, while agents that have a vapor density lower than the density of air, e.g. hydrogen cyanide, dissipate into the atmosphere. As a result, when deployed outdoors on the battlefield, chemical warfare agents that have a vapor density higher than the density of air are more persistent than those that have a lower vapor density (43,44). To be sure, vapor density in not the only parameter that determines the persistency of chemical warfare agents. In particular, persistency is greatly affected by the propensity of the agents to degrade, where more stable agents, e.g. sulfur mustard, with a lower tendency to degrade have a higher persistency than less stable agents.
Water solubility, together with reactivity, directly affects the interaction of inhaled chemical warfare agents with the lungs. In particular, chemicals that are more water-soluble and/or are more reactive, e.g. hydrochloric acid, will damage the upper airways. Conversely, chemicals that are less water soluble and reactive, such as phosgene or perfluoroisobutylene (PFIB), will penetrate deeply into the lungs and damage the lower airways. Chemicals with intermediate water solubility, such as chlorine, will damage both the upper and the lower airways (45). We will come back to this point later on, in Section 4.2. On the other hand, lipophilicity, i.e. higher affinity for greasy substances than water, favors the absorption of toxic chemicals by the skin. For instance, lewisite, which is more lipophilic than sulfur mustard, is absorbed by the skin more readily than the latter and its toxic effects have a significantly shorter onset time (see Section 4.1).
A number of physicochemical properties – chiefly lipophilicity, polar surface area, and molecular weight – affect the ability of chemical warfare agents to penetrate the blood-brain barrier (BBB), a physical barrier posed by the cells lining the walls of the capillaries in the brain and the spinal cord. In turn, the ability to penetrate the blood-brain barrier determines the ability or lack thereof of chemical warfare agents – as well as the pharmacological countermeasures against them – to exert their effect on the central nervous system (CNS). Small hydrophobic molecules tend to cross the blood-brain barrier by passive diffusion, while large hydrophilic molecules do not. Of note, entering and remaining in the central nervous system is a complex issue that goes beyond passive diffusion, involving also active transport, a process that depends on molecular recognition between the chemicals and the macromolecular transporter. In particular, some transporters allow select molecules to cross the blood-brain barrier, while others move back outside of the central nervous system molecules that have passively crossed the blood-brain barrier (46,47).
Table 1. Physicochemical properties and toxicity data of selected chemical warfare agents. Toxicity values refer to humans, unless otherwise noted.
| Chemical | M.P. (°C) | B.P. (°C) | Vapor Pressure (mm Hg) | Vapor Density (Air = 1) | Water solubility (mg/L) | LD50 (mg/kg) | LCt50a (mg/m3) min | Ref. |
| Vesicants | ||||||||
| Sulfur mustard (HD) | 13-14 | 227 | 0.072 at 20°C | 5.4 | 920 at 22°C | 100 (skin) | 1500 | (48,49) |
| Mechlorethamine (HN2) | -60 | 75 | 0.17 at 25°C | 5.9 | Very slight | 29b (skin) | 3000 | (50,51) |
| Lewisite | 0.1 | 190 | 11.2 at 25°C | 7.1 | Slight | 40-50 (skin) | 1200-1500 | (48,52) |
| Choking agents | ||||||||
| Chlorine | -101.5 | -34.04 | 5830 at 25°C | 2.48 | 6300 at 25°C | NAc | 13500d | (53) (54) |
| Phosgene | -118 | 8.2 | 1420 at 25°C | 3.4 | Slight | NAc | 1500e | (55) (37) |
| PFIB | -130 | 7 | 1740 at 25°C | 6.3 | NAc | NAc | 1031f,g | (56) |
| Chloropicrin | -64 | 112 | 24 at 25°C | 5.7 | 1900 at 20°C | 37.5g (oral) | 23234g,h | (57) |
| Chemosensory irritants | ||||||||
| CN | 55.6 | 247 | 5.5×10-3 at 20°C | 5.3 | 1805 at 25°C | 127g (oral) | 8750g,i ICt50: 20-40 | (40,58,59) |
| CS | 95-96 | 310-315 | 3.4×10-5 at 20°C | 6.5 | Sparingly | 1366g (oral) | 88840g,i ICt50: 3-10 | (40,59,60) |
| CR | 73 | NAc | 2.2×10-4 at 20°C | 6.7 | 124 at 25°C | 7500g (oral) | 139000g,j ICt50: 1 | (40,59,61) |
| Capsaicin | 65 | NAc | 1.3×10-8 at 25°C | NAc | 28.93 at 25°C | 190g (oral) | 835000 | (40,62) |
| Blood agents and related toxic chemicals | ||||||||
| Hydrogen cyanide | -13.28 | 25.7 | 740 | 0.99 | Complete at 25°C | 100 (skin) | 2500-5000 | (63,64) |
| Cyanogen chloride | -6.55 | 12.9 | 1000 | 2.1 | 69000 at 20°C | 100 (skin) | 11000 | (63,65) |
| Arsine | -116 | -62.5 | 11000 at 20°C | 2.66 | 280 at 20°C | 2.5b,k | 1200b,l | (66) |
| Hydrogen sulfide | -85.49 | -60.33 | 13600 at 20°C | 1.189 | 3980 at 20°C | NAc | 27000g,m | (67) |
| Carbon monoxide | -205.02 | -191.5 | 1.55×108 | 0.968 | Sparingly | NAc | 586560b,h | (68) |
| Nerve agents | ||||||||
| Tabun (GA) | -50 | 247 | 0.07 at 25°C | 5.6 | 98000 at 25°C | 14.3 | 150-400 | (69,70) |
| Sarin (GB) | -56 | 147 | 2.9 at 25°C | 4.86 | Miscible | 24.3 | 70-100 | (69,70) |
| Soman (GD) | -42 | 167 | 0.3 at 25°C | 6.3 | 21000 at 25°C | 5 | 35-50 | (69,70) |
| VX | -39 | 300 | 0.0007 at 25°C | 9.2 | Miscible | 0.14 | 10-30 | (69,70) |
| VR (R-33) | NAc | 323 | 0.0003 at 25°C | NAc | NAc | 0.1-0.01 | 40 | (69,70) |
| Central incapacitating agents | ||||||||
| 3-Quinuclidinyl benzilate (BZ) | 167.5 | 320 | 2.38×10-10 at 25°C | 11.6 | 200 at 25°C | 0.25o ID50: 0.0062n,o | 3700i,p ICt50: 110q | (71-73) |
| Fentanyl | 85.2 | NA | 4.43×10-9 at 25°C | NAc | 200 at 25°C | 11.2b,r ED50: 0.0014b,r,s | 528b,i,t | (74-76) |
a Inhalation
b Mouse data.
c Data not available in the cited references.
d LCt50 for inhalation for a 2 min exposure is estimated at about 13500 and 9500 (mg/m3) min for military personnel and general population, respectively.
e Extrapolated from LC50 for a 2 min exposure.
f Extrapolated from LC50 for a 2 h exposure.
g Rat data.
h Extrapolated from LC50 for a 4 h exposure.
i Aerosol.
j Pyrotechnically generated smoke.
k Intraperitoneal injection.
l Extrapolated from LC50 for a 2.4 min exposure.
m Extrapolated from LC50 for a 18 min exposure.
n Intramuscular injection.
o Incapacitating central effects.
p For general public.
q Military incapacitation.
r Intravenous injection.
s Antinociception.
t For a 1 h exposure.
4. Classes of chemical warfare agents.
For convenience, chemical warfare agents can be grouped into categories endowed with common characteristics. Among the criteria that are used to classify chemical warfare agents, the signs and symptoms caused by the agents’ toxicity occupy a prominent place. Signs are objective manifestations of a syndrome that are apparent to an external observer (e.g. restriction or dilation of the pupils). Conversely, symptoms are subjective manifestations of a syndrome that can only be described by the affected person (e.g. feeling pain). Grouped together, signs and symptoms of a chemical intoxication are also known as toxidromes, from the fusion of the words toxic and syndrome (77,78).
According to their toxidromes, chemical warfare agents can be classified into:
- Vesicants, or blister agents, which derive their name from their primary action on the skin;
- Choking agents, or lung damaging agents, which derive their name from their primary action on the airways;
- Irritants, including but not limited those used for riot control, which derive their name from their ability to induce irritation to the skin, the eyes, or mucous membranes.
- Blood agents, or knockdown agents, which derive their name from their ability to prevent the organism from utilizing oxygen for aerobic metabolisms, thus eventually leading to coma;
- Cholinergic agents, including but not limited to nerve agents, which derive their name from their ability to overstimulate cholinergic neurotransmission through the inhibition of the enzyme acetylcholinesterase;
- Centrally incapacitating agents, also referred to as Central Nervous System (CNS)-Acting Chemicals, which comprise a variety of chemicals endowed with a wide range of effects on the central nervous system, such as opioids (opioid toxidrome) or anticholinergic substances (anticholinergic toxidrome).
Below, we will review the above-mentioned classes of chemical warfare agents, illustrating their structures, physicochemical characteristics, mechanisms of action, and medical countermeasures. For each class, we will delve more deeply into the specifics of one or more representative chemical warfare agents, as a case study. We will concentrate exclusively on synthetic toxic chemicals. For biological toxins, which, as mentioned, are toxic chemicals produced by leaving organisms and endowed with disparate toxidromes, the reader is referred to other sources (16-18).
4.1. Vesicants. Vesicants, also known as blister agents, are low volatility substances that attack primarily the skin. Other prominent targets of vesicants include the eyes and the airways. Chemical warfare agents typically classified as vesicants include, among others, sulfur mustards, nitrogen mustards, lewisites (Fig. 2). Phosgene oxime (Fig. 2) is also considered a vesicant by the U.S. military, although it does produces solid lesions resembling hives rather than blisters. Beyond chemical warfare agents, a number of industrial chemicals share toxidromes with vesicants. Chiefly, these include corrosive substances such as strong acids or bases as well as strong oxidants (48,78).
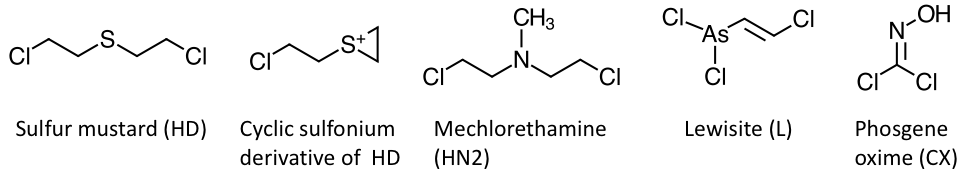
Fig. 2. Examples of vesicants. The highly reactive cyclic sulfonium derivative of HD is also shown.
Sulfur mustards are the prototypical examples of vesicant agents. They are thioether molecules featuring chloroalkyl substituents, which were first synthesized in the 1800s. Among them, the most common is bis(2-chloroethyl)sulfide, codenamed HD, and frequently simply called sulfur mustard or Yperite, as it was first used in battle by the Germans in World War I (WWI) on July 12, 1917, during the course of the Third Battle of Ypres, in Belgium. Sulfur mustard is often referred to as mustard gas. However, this is a misnomer, as, with a boiling point of 227ºC, sulfur mustard is an oily liquid at room temperature. In its vapor form, mustard has a vapor density about 5.5 times higher than the density of air. Thus, upon evaporation, sulfur mustard vapors stay low to the ground without dissipating into the atmosphere. Sulfur mustard has an LD50 of 100 mg/kg for cutaneous exposure to the liquid agent and an LCt50 of 10000 (mg/m3) min for inhalation. It is considered a persistent chemical warfare agent, as it can last from days to years in soil, depending on the temperature. As we explain in greater detail in the next section, sulfur mustards, and mustards in general, are alkylating agents and their mechanism of action is based on the alkylation of nucleic acids and proteins (48,79,80).
Nitrogen mustards were synthesized in the 1930s but were not used in combat. Interestingly, in the early years of chemotherapy, one nitrogen mustard (mechlorethamine, or mustine, codenamed HN2) was successfully developed into an effective chemotherapy agent, especially for the treatment of leukemia (48,79,80). The development of HN2 into a chemotherapy drug was prompted by medical observations following the Bari incident in December 1943, when an aerial attack by the Germans struck a U.S. ship carrying sulfur mustard anchored at the southern Italian city. Victims exposed to sulfur mustards as a consequence of the incident were found to have a particularly low count of leukocytes, thus stimulating the medical community to conduct studies for the development of mustards into anti-leukemia drugs (81).
Lewisites are arsenic-containing compounds that exert their toxic properties by reacting with thiol groups of proteins. As a result, they inhibit a number of enzymes including, among others, some key enzymes in the carbohydrate metabolism. The prototypical lewisite is b-chlorovinyldichloroarsine), codenamed L and often simply referred to as lewisite from the name W. L. Lewis, a U.S. Army Captain whose research team developed it into a chemical warfare agent during WWI. Lewisite has a boiling point of 190ºC and a vapor density 7.1 times higher than the density of air. It is endowed with an LD50 of 40 to 50 mg/kg for cutaneous exposure to the liquid agent and an LCt50 of 1200-1500 (mg/m3) min for inhalation, thus being somewhat more potent than sulfur mustard. Of note, lewisite is substantially more hydrophobic than sulfur mustard. Its toxic effects on the skin, the lungs and the eyes are similar of those of sulfur mustard. However, unlike for sulfur mustard, its effects have a rapid onset, becoming manifest just a few minutes after exposure. An antidote against lewisite, the “British antilewisite”, also known as BAL, has been developed (Fig. 3). This antidote, which has been shown to be effective if used soon after exposure to the agent, contains thiol groups and reacts with the arsenic atom of lewisite before the chemical warfare agent gets the chance to interact with its protein targets (48,80).

Fig. 3. Dimercaprol, the “British antilewisite” antidote (BAL).
4.1.1 Sulfur mustards. Sulfur mustards are alkylating agents. After cyclization and loss of a chlorine ion, sulfur mustards form a strongly electrophilic and highly reactive cyclic sulfonium ion (Fig 2). The most studied molecular target of sulfur mustards alkylation is deoxyribonucleic acid (DNA). In particular, sulfur mustards have been shown to alkylate purine basis, mostly at the level of the N7 nitrogen of guanosine and, to a minor extent, the N3 nitrogen of adenine. Thiol groups in proteins and peptides are also prominent molecular targets of sulfur mustards. Notably, the alkylation of a methionine residue in human serum albumin, Met329, has been recently identify as a target for alkylation by sulfur mustard, useful as a bioanalytical marker (82,83).
Sulfur mustards penetrate within the skin, causing, with a delayed onset, erythema and the formation of vesicles at the level of the dermal-epidermal junction, the area where the most external layers of the skin, known as epidermis, join the deeper layers, known as dermis. Erythema usually appears after 2 to 24 hours following exposure and vesicles form several hours later. Mustard vapors cause acute inflammation of the upper and lower airways, through inhalation, as well as an array of dose-dependent effects on the eyes, ranging from irritation to corneal damage (48,79,80,82).
Several hypotheses have been advanced on the molecular mechanism of action of sulfur mustards, including the poly(ADP-ribose) polymerase (PARP) hypothesis, the thiol depletion hypothesis, and the lipid peroxidation hypothesis. The PARP hypothesis is based on the reactivity of mustard with DNA. In particular, the breakage of DNA strand consequent to the alkylation of purine bases by sulfur mustards causes the activation of PARP enzymes, which in turn leads to cytotoxic effects that culminate in apoptosis or necrosis. The thiol depletion and lipid peroxidation hypotheses are both based on the reactivity of sulfur mustards with thiol groups. In particular, they are based on the reactivity of sulfur mustards with the peptide glutathione (GSH), a tripeptide containing a cysteine residue. Importantly, GSH contributes to both keeping the thiol groups of proteins in the reduced state and contrasting the action of reactive oxygen species. According to the thiol depletion hypothesis, reduced GSH levels lead to the inactivation of enzymes known as Ca2+/Mg2+ ATPases due to the loss of free thiol groups. In turns, the inactivation of such enzymes leads to an abnormal increase of the intracellular calcium levels, consequently leading to the activation of enzymes that degrade proteins, nucleic acids, and lipids, thus culminating in cell death. Lastly, according to the lipid peroxidation hypothesis, reduced GSH levels cause an increase in the concentration of hydrogen peroxide, with the consequent formation of toxic oxidants that react with phospholipids in cellular membranes and the eventual breakage of the membranes (48,84). Beyond their action at the dermal-epidermal junction, sulfur mustards can also have serious systemic effects. Chiefly, these include bone marrow depression, which leads to the suppression of the immune system (48,82).
In the absence of antidotes and specific pharmacological options, the treatment of victims of exposure to sulfur mustards generally aims at preventing infections through the use of antibiotics, reducing inflammation, e.g with anti-inflammatory drugs, including non-steroidal anti-inflammatory drugs (NSAIDs), and in the case of airways exposure, facilitating breathing. Although specific treatment options for exposure to sulfur mustards are currently unavailable, the increased understanding of the mechanisms of action underlying their toxicity has led to the investigation of several potential therapeutic approaches. Among others, possible options include PARP inhibitors, inhibitors of apoptosis-mediating caspases, substances that can reduce neurogenic inflammation, or a peptide isolated from iodine-treated burns (82).
Prompt decontamination with soapy water and possibly dilute bleach (hypochlorite) is essential to prevent the damages caused by mustard agents. Of note, mustards are particularly insidious agents, because, although their effects are delayed, prompt decontamination is essential to reduce or avoid damage (48). This is well illustrated by the case of a Syrian family who was exposed to sulfur mustard when the city of Marea was attacked by non-state actors in August 2015. Failure to promptly decontaminate or change clothes (only the father showered with just water) led to the appearance of skin blisters about 10 hours or longer after exposure (85).
4.2. Choking agents. Choking agents, also known as lung damaging agents, are high volatility substances that attack primarily the airways. Chemical warfare agents typically classified as choking agents include chlorine, phosgene, perfluoroisobutylene (PFIB), and chloropicrin among others (Fig. 4). Due to their volatility, choking agents are non-persistent chemical warfare agents. However, choking agents typically have vapor densities higher than the density of air. Hence, after deployment, they stay close to the ground and do not readily dissipate. Beyond chemical warfare agents, a number of volatile industrial chemicals share toxidromes with choking agents, such as sulfur dioxide, ammonia, and formaldehyde, among others (42,78,86). Chloropicrin is an exception in terms of volatility with respect to other choking agents, with a boiling point of 112ºC and a vapor pressure of 24 mm Hg at 25ºC. Chloropicrin differs from canonical choking agents also for its toxidromes, having toxic effects that are intermediate between those of choking agents and irritating harassing agents. Consequently, it is often classified in the latter category (57,87).

Fig. 4. Examples of choking agents.
Upon inhalation, choking agents damage cells and tissues in the respiratory system, causing inflammation and necrosis. As mentioned, water solubility and reactivity play a key role in determining the section of the respiratory tract that will be affected the most by a specific choking agent. Chemicals that are more water-soluble and/or are more reactive target primarily the upper airways (i.e. the central compartment of the respiratory system). Conversely, chemicals that are less water soluble and/or less reactive penetrate more deeply into the lungs and damage the lower airways (i.e. the peripheral compartment of the respiratory system). Damage to the upper airways caused by centrally acting choking agents typically leads to swelling and restriction of breathing. Damage to the lower airways caused by peripherally acting choking agents typically results in pulmonary edema. Treatment of exposure to choking agents is typically supportive, and includes ventilation support, fluid replacement, and anti-inflammatory drugs. Anti-inflammatory lipid mediators that block the production of leukotrienes and prostaglandins have been shown to be effective as post-exposure drugs to counteract the toxic action of phosgene, resulting in less pulmonary edema (42,86,88).
4.2.1 Chlorine and phosgene. Chlorine is a greenish gas with a boiling point of -34 ºC and a vapor pressure of 5830 mm Hg at 25ºC. Its vapor density is 2.5 times higher than the density of air. Its LCt50 for inhalation for a 2 min exposure is estimated at about 13500 and 9500 (mg/m3) min for military personnel and general population, respectively. However, chlorine does not follow Haber’s law and has a toxic load exponent higher than 1, i.e. the agent’s concentration has a higher impact than the duration of exposure on chlorine’s toxic effects. Notoriously, chlorine gas was employed as a chemical warfare agent during WWI by the Germans during the Second Battle of Ypres, on April 1915, leading the way to a massive use of lethal chemical warfare agents by both sides during the conflict. Chlorine has medium water solubility (6.3 g/L). As a result, it exerts toxic effects on both the upper and lower airways, which appear almost immediately after exposure (42,53,54,86,88).
Phosgene is a volatile chemical with a boiling point of 8.2ºC and a vapor pressure of 1420 mm Hg at 25ºC. Its vapor density is 3.4 times higher than the density of air. Its LCt50 for inhalation is about 2000 (mg/m3) min. However, Haber’s law is applicable to phosgene only at very low concentrations and, in general, just like chlorine, phosgene has a toxic load exponent higher than 1. Phosgene, which has numerous large-volume applications in the chemical and pharmaceutical industry, was first weaponized during WWI and was largely used by both sides. It is poorly soluble in water. As a result, it is a peripherally acting chemical warfare agent that exerts its effect on the lower airways, where it causes damage to the alveolar capillaries leading to pulmonary edema and consequent difficulties in breathing that can culminate in respiratory arrest. Phosgene has also been shown to lead to reduced GSH levels, with consequent oxidative stress. The toxic effects of phosgene are delayed, with an onset typically occurring between 1 hour to 1 day after exposure (42,55,86,88).
4.3. Irritants. Irritants are chemicals that cause irritation to the skin, exposed mucous membranes, eyes, and airways. They are also referred to as harassing agents. Chemical warfare agents belonging to this category include, among many others, acrolein, ethylbromoacetate, bromoacetone, and the above-mentioned chloropicrin (Fig. 5). Of note, ethylbromoacetate as well as other harassing chemicals were amply used on the battlefield during the early part of WWI by both parties, prior to the beginning of the employment of lethal choking agents and vesicants (31,40,87,89).

Fig. 5. Examples of irritant chemical warfare agents.
Less toxic irritants are employed by law enforcement as riot control agents for crowd control purposes. As explained above, the CWC defines riot control agents as “any chemical not listed in a Schedule, which can produce rapidly in humans sensory irritation or disabling physical effects which disappear within a short time following termination of exposure” (CWC Article II, paragraph 7) (1). The key here is that the effects should have a rapid onset and should not be long lasting. Hence, any chemical that causes prolonged or permanent damaged is excluded by this definition. Above all, as already noted, to be considered for use as riot control agents, chemicals must exert their harassing effect at doses and concentrations that are significantly lower than those that would cause lethality or long-lasting harm, so as to grant their safe employment for law enforcement purposes. As mentioned, the OPCW SAB identified 17 chemicals that, based on the toxic effects resulting from acute exposure under appropriate usage conditions (i.e. “in outdoor scenarios where the chemicals would be present in low dilution in air, in non-enclosed spaces, with the people affected able to escape unimpeded from the irritant cloud”), fit the CWC definition of the riot control agents. Most of these are peripheral chemosensory irritants (PCSI). Although PCSI riot control agents are typically in the solid state at room temperature, they are often referred to as tear gasses as they are frequently dispersed into the air in an aerosolized form (31).
4.3.1 Peripheral chemosensory irritant riot control agents. Peripheral chemosensory irritants are chemicals that exert their action by activating two chemosensory receptors in the peripheral nervous system (PNS) belonging to the superfamily of transient receptor potential (TRP) channels: the transient receptor potential ankyrin 1 (TRPA1) or the transient receptor potential vanilloid 1 (TRPV1). TRPs are cation channels that play a key role in sensory physiology. In particular, the TRPA1 and TRPV1 receptors are activated by cold and heat, respectively. Moreover, they are both activated by a wide range of substances of botanical origin, including, among others, the isothiocyanates contained in mustard oil, horseradish or wasabi for the TRPA1 receptor, and the vanilloids contained in chili peppers for the TRPV1 receptor (40,87,90).
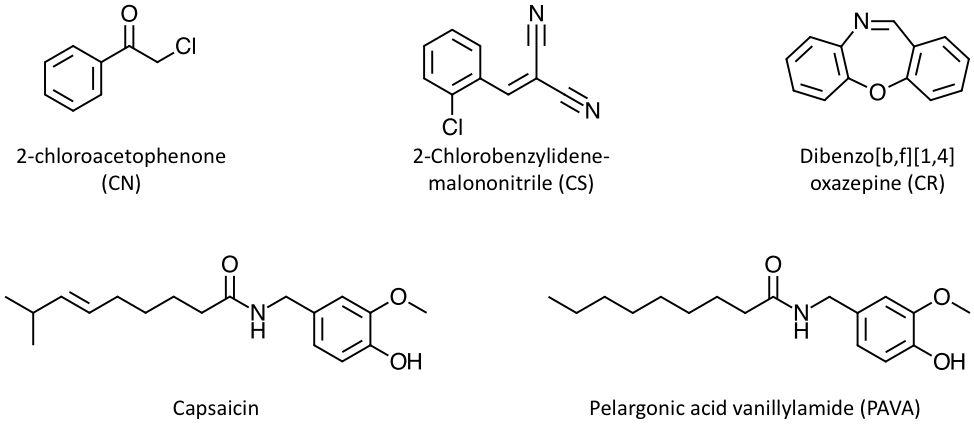
Fig. 6. Examples of chemosensory irritant riot control agents.
Riot control agents that function through the activation of the TRPA1 receptor include 2-chloroacetophenone (CN) 2-chlorobenzylidenemalononitrile (CS), and dibenzo(b,f)[1,4]-oxazepine (CR) (Fig. 6). The first one was first synthesized in Germany in the 1860s. The other two were first synthesized in the United Kingdom, in the 1920s and 1960s, respectively, seeking increased incapacitating potency, lower lethality, and higher stability. Of note, CS has a lethality lower than that of CN, to which it is preferred as a riot control agent in several countries. However, CS is a very stable compound, which makes it persistent in the environment. This family of riot control agents can be dispersed through a variety of means, including pyrotechnic smokes, hand grenades, mortar shells, or aerosols. They can also be dispersed in solution as liquid jets or sprays (40,87,89).
Riot control agents that function through the activation of the TRPV1 receptor include oleoresin capsicum (OC), capsaicin, and pelargonic acid vanillylamide (PAVA) (Fig. 6). Oleoresin capsicum is not a pure substance, but it is an oily resin containing a mixture of vanillamides, derived from the fruits of plants belonging to the Capsicum genus, which include chili peppers. Conversely, capsaicin is a pure substance and is the main active component of oleoresin capsicum, amounting to about 70% of the vanillamides that can be found in the resin. PAVA is a synthetic derivative of capsaicin, which features a simpler linear chain with no double bonds or branches. These riot control agents are typically dispersed as sprays, with a variety of carriers and propellants. Of note, OC-based sprays are also legally available to the general public for personal protection in many countries (40,87,89).
4.4. Blood agents. Blood agents are chemical warfare agents that interfere with the ability of the body to utilize oxygen for aerobic cellular metabolisms. The most common chemical warfare agents classified as blood agents include cyanides and arsine (Fig. 7). Among cyanides, hydrogen cyanide, codenamed AC, and cyanogen chloride, codenamed CK, are volatile chemicals, with a boiling point of 25.7 and 12.9ºC and a vapor pressure of 740 and 1000 mm Hg, respectively. Hence, they are both non-persistent chemical warfare agents. However, the two chemicals differ significantly in terms of their vapor density: while hydrogen cyanide has a vapor density lower than the density of air (0.99), cyanogen chloride’s vapor density is 2.1 times the density of air. Thus, when deployed on the battlefield, the former tends to dissipate into the atmosphere, while the second one does not. While ineffective on the battlefield, hydrogen cyanide can reach very lethal concentrations when used indoors. In the 1920s, Fritz Haber developed a formulation of hydrogen cyanide known as Zyklon B, intended to be used as a pesticide. Notoriously, during the Holocaust, the Nazis used Zyklon B in gas chambers at extermination camps. Cyanide salts, e.g. potassium cyanide, are solids at NTP. Arsine is a highly volatile, nonpersistent chemical warfare agent, with a boiling point of -62.5ºC and a vapor pressure of 11000 mm Hg at 20ºC. Its vapor density is higher than the density of air (44,63,66,91).

Fig. 7. Examples of blood agents.
Despite their name, not all blood agents affect the ability of blood to carry oxygen to the tissues. However, although through different mechanisms of action, they all prevent cells from conducting aerobic metabolisms. Hence, from the toxidromes perspective, blood agents can be classified as asphyxiating chemicals. Of note, due to the severity and the rapid onset of the symptoms, the asphyxiating chemicals toxidrome is also referred to as “knockdown” toxidrome. By preventing cells from catabolizing dietary fuel aerobically, blood agents severely impair the ability of cells to synthesize adenosine triphosphate (ATP), which is the energy currency utilized in cellular processes that require energy (endergonic processes). Consequently, cells switch to anaerobic glycolysis to synthesize ATP, with the resulting production of lactic acid, thus leading to acidosis. Low ATP levels and acidosis have severe effects on the organism, affecting chiefly the nervous and the cardiac system, leading to coma, seizures, and cardiorespiratory crisis (44,63,78,91).
Cyanides block the last step of the respiratory chain, thus preventing cells from utilizing oxygen for aerobic metabolism, by inhibiting the enzyme complex cytochrome c oxidase. Arsine destroys red blood cells, thus depriving cells of oxygen, without which aerobic metabolisms cannot be conducted. Beyond chemical warfare agents, other asphyxiating chemicals include hydrogen sulfide, carbon monoxide, carbon dioxide, and methane. Hydrogen sulfide has the same mechanism of action of cyanides, based on the inhibition of cytochrome c oxidase. Carbon monoxide’s mechanism of action is based on its very high affinity for the heme prosthetic group of hemoglobin and myoglobin, significantly higher than the affinity of oxygen for the same group. By binding very tightly to hemoglobin’s heme, carbon monoxide prevents hemoglobin from binding oxygen and carrying it, through the bloodstream, from the lungs to the tissues. Conversely, carbon dioxide and methane simply displace oxygen from the atmosphere, thus preventing physiological respiration (78).
4.4.1 Cyanides. Cyanides exert their toxicity by binding to the heme prosthetic group of the enzyme complex cytochrome c oxidase, also known as complex IV, beyond binding to other heme-containing proteins as well (44,92). Complex IV is the last macromolecular complex involved in the respiratory chain, a mitochondrial process in which reducing agents produced in the course of the aerobic catabolism of dietary fuels get re-oxidized while their electrons, through a number of steps involving several electron carriers, eventually land on molecular oxygen that gets reduced to water. As this happen, protons are syphoned out of the mitochondrial matrix, thus creating an electrochemical gradient and a resulting “proton-motive force,” which is ultimately utilized to synthesize ATP as the protons reenter the mitochondrial matrix flowing through the enzyme ATP synthase. By inhibiting the activity of Complex IV, cyanides block the entire respiratory chain, thus preventing the aerobic production of ATP, with the above-mentioned consequences.
Hydrogen cyanide has an LD50 value for liquid exposure through the skin of around 100 mg/kg. For inhalation, hydrogen cyanide has an LCt50 ranging from 2500 to 5000 (mg/m3) min, depending on the time of exposure, with a toxic load exponent higher than 1. The toxic exponent higher than 1, i.e. the stronger impact of concentration than duration of exposure on the effects of cyanide, are due to the fact that the human body is capable of detoxifying low levels of cyanide. Humans are naturally exposed to cyanide through a number of cyanogenic foods, including several fruits and vegetables. Tobacco smoke is a source of cyanide as well. However, as mentioned, the human body is equipped with several natural mechanisms for the detoxification of cyanide, chiefly through its conversion into the less toxic chemical thiocyanate (SCN), mediated by rhodanese and a number of additional enzymes. Other mechanisms of detoxification include the binding of cyanide to methemoglobin (hemoglobin in which the iron ion is oxidized to the Fe+3 state), leading to cyanomethemoglobin, and to vitamin B12, or cobalamin, leading to the formation of cyanocobalamin. Antidotes for cyanide poisoning are meant to boost these detoxification mechanisms and include sulfur donors to enhance the sulfurtransferases, such as sodium thiosulfate, chemicals that enhance the formation of methemoglobin, such as sodium nitrite, the injectable form of vitamin B12 hydroxocobalamin, and other cobalt-containing molecules that can directly bind cyanide (44,63,91).
4.5. Cholinergic agents. Cholinergic agents are chemicals that increase the synaptic concentration of the neurotransmitter acetylcholine by inhibiting the activity of the enzyme acetylcholinesterase. The most prominent chemical warfare agents belonging to the cholinergic toxidrome are nerve agents, organophosphorus chemicals that act as irreversible inhibitors of acetylcholinesterase. Beyond nerve agents, the cholinergic toxidrome includes also other acetylcholinesterase inhibitors, such as organophosphorus and carbamate pesticides (78).
Acetylcholine is a key neurotransmitter that plays a plethora of functions in both the peripheral and the central nervous system, through the activation of two classes of receptors known as muscarinic and nicotinic acetylcholine receptors. Muscarinic receptors belong to the superfamily of G protein-coupled receptors (GPCRs), while nicotinic receptors are ligand-gated ion channels, or ionotropic receptors. After being secreted by presynaptic vesicles, acetylcholine exerts its action by activating presynaptic and postsynaptic acetylcholine receptors. However, soon after its secretion, acetylcholine gets degraded into choline and acetic acid by acetylcholinesterase, thus ensuring the termination of the signal. Choline is then transported back into the presynaptic neurons, where it is converted back to acetylcholine. The inhibition of acetylcholinesterase by cholinergic agents leads to a prolonged overstimulation of muscarinic and nicotinic acetylcholine receptors, causing at first an overwhelmingly high activity of cholinergic neurotransmission (i.e. the neurotransmission mediated by acetylcholine), followed by its suppression due to the desensitization of the acetylcholine receptors. Physiological functions affected by altered cholinergic transmission include the “rest-and-digest” function mediated by the parasympathetic nervous system, the “fight-or-flight” reaction mediated by the sympathetic nervous system, the activity of motor nerves of the somatic nervous system, and a wealth of functions of the central nervous system. Due to the multiple physiological roles of acetylcholine, many signs and symptoms are associated with the cholinergic toxidrome, including, among several others, increased secretions, bladder and bowel incontinence, vomiting, constriction of the pupil (miosis), muscle twitching, paralysis, mental-status changes, and coma. Respiratory failure and seizures are common causes of death (69,70,77,78,93-95).
4.5.1 Nerve Agents. As mentioned, nerve agents are organophosphorus chemicals that act as irreversible inhibitors of acetylcholinesterase. Specifically, nerve agents form a stable covalent bond with a serine residue in the enzyme’s active site (Ser203 in the human enzyme), which is not easily hydrolysable. The serine residue to which nerve agents bind is fundamental for the catalytic activity of acetylcholinesterase. Hence, upon binding nerve agents, the enzyme loses its ability to breakdown acetylcholine. Some oximes are capable of reacting with the bound nerve agents, thus cleaving the agent from the enzyme and leading to the reactivation of the latter. However, after a period of time that depends on the specific nerve agent, acetylcholinesterase-bound nerve agents undergo a dealkylation process known as aging. To date, there are no known oximes that can reactivate acetylcholinesterase-nerve agent adducts after aging (69,70,93).
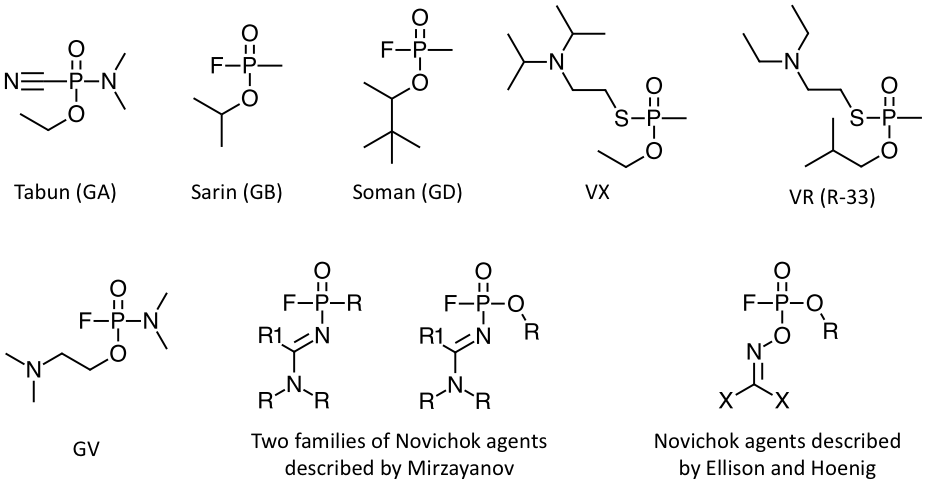
Fig. 8. Examples of nerve agents. In Mirzayanov’s Novichok structures, the R groups stand for alkyl groups, while the R1 group stands for alkyl groups or a tertiary amine (when R1 is an alkyl group, the compounds are amidine derivatives; when R1 is an amine, the compounds are guanidine derivatives). In the Ellison and Hoenig’s Novichok structures, the R group stands for a variety of halogenated alkyl groups, while the X groups stand for halogens.
Several generation of nerve agents have been developed throughout the years. The first nerve agents, known as G agents (from Germany), were developed in Germany prior to and during World War II (WWII). Among others, G agents include tabun (GA), sarin (GB), and soman (GD) (Fig. 8). After the war, a second generation of nerve agents, known as V agents, was developed by the Western Bloc and, soon after, the Soviet Union as well. Prototypical V agents include VX, which was firstly synthesized in the United Kingdom and subsequently developed in the United States, and its Soviet counterpart VR, also known as R-33 (Fig. 8). V agents are less volatile and more persistent that G agents. For instance, the vapor pressure at 20°C is 2,1 mm Hg for sarin and 0.0007 mm Hg for VX. The persistence of sarin in soil is from 2 to 24 hours, while that of VX is from 2 to 6 days. A third generation of nerve agents, known as intermediate volatility agents (IVA), an example of which is GV, was developed in the United States later in the Cold War. Finally, a fourth generation of nerve agents, also known as Novichok agents or A-agents, was developed in the Soviet Union beginning in the 1970s. Novichok agents have been described as very persistent in the environment and, reportedly, have potencies similar to that of VX and sometimes higher (although data confirming these statements are not publicly available). Several families of nerve agents are comprised within the umbrella of the Novichok definition. Two related families of Novichok agents have been described by whistleblower Vil Mirzayanov in a 2009 book (Fig. 8). Other structures for Novichok agents had been previously reported by Ellison and Hoenig (Fig. 8) (10,69). For a variety of nerve agents, binary weapons were developed as well, based on two less toxic precursors that, when combined together, yielded a lethal nerve agent (69,70,93,95,96).
Nerve agents were not used in WWII. However, in the 1980s, they were used by Iraq against Iranian forces and against the Kurds. In 1994 and 1995, sarin was used against civilians in terrorist attacks in Japan by the cult Aum Shinrikyo. Nerve agents were subsequently used in a series of attacks in the course of the Syrian civil war, the largest one of which took place in August 2013 in the Ghouta region, causing the death of 1400 people. In February 2017, Kim Jong-nam, the half-brother of North Korea’s leader Kim Jong-un, was assassinated with a binary VX agent at the Kuala Lumpur airport, in Malaysia. About a year later, in March 2018, a Novichok agent was deployed in Salisbury, UK, for the attempted assassination of a former Russian intelligence officer, Sergei Skripal, who had defected to the British MI6. A few months after the attack, a perfume bottle containing the same Novichok agent was found in the area, leading to the poisoning and the subsequent death of a women who sprayed herself with what she thought was genuine perfume. It is worth noting that a proposal to add chemicals related to the agent deployed in Salisbury to Schedule 1 of the CWC, jointly submitted to the OPCW by the United States, Canada, and the Netherlands, clarified that the chemical used in the assassination attempt belongs to one of the Novichok families described by Vil Mirzayanov (6,10,11,30,69,70,96,97).
The standard treatment of nerve agent poisoning is based on a three-pronged strategy that includes the administration of an antagonist of the muscarinic acetylcholine receptors (e.g. atropine), an oxime reactivator (e.g. pralidoxime, also known as 2-PAM), and a benzodiazepine (e.g. diazepam). Muscarinic antagonists are meant to contrast the activation of muscarinic acetylcholine receptors by the elevated acetylcholine levels, thus lowering the muscarinic symptoms of nerve agent poisoning. Oxime reactivators are meant to cleave the bound nerve agent off the acetylcholinesterase enzyme, thus restoring the body’s ability to degrade acetylcholine. Benzodiazepines, which are positive allosteric modulators of the ionotropic GABAA receptors, are meant to prevent convulsive seizures. A kit developed by the U.S. Armed Forces and known as ATNAA (antidote treatment nerve agent auto-injector), has all three components. Moreover, pretreatment options have been developed with the intent of providing protection to those who are at risk of exposure to nerve agents. The U.S. Food and Drug Administration (FDA) has approved the use of pyridostigmine, a reversible acetylcholinesterase inhibitor, as a pretreatment option. By binding reversibly to the enzyme’s catalytic site, pyridostigmine makes it less available to nerve agent binding, thus providing protection (69,70,93,98,99).
A great deal of research is ongoing to develop improved treatment and pretreatment options for nerve agent poisoning, some of which is highlighted below. Novel oximes are being researched with the intent of developing reactivators that can provide equal protection against a broad spectrum of nerve agents and can better penetrate the blood-brain barrier, thus yielding better reactivation of the enzyme within the central nervous system. Improved small molecule pretreatment options are highly sought after as well. Among them, galantamine, a reversible acetylcholinesterase inhibitor and a potentiating activating ligand of the nicotinic receptors, appears particularly promising. It is more permeable to the blood-brain barrier than pyridostigmine, thus providing better protection to the central nervous system. Moreover, its activity at the nicotinic receptors is likely to counteract the loss of nicotinic signaling due to the desensitization of the nicotinic receptors that follows their overstimulation. Several protein-based pretreatment options are under development as well. These include stoichiometric bioscavengers, which sequester nerve agents before they bind to acetylcholinesterase, and catalytic bioscavengers, which break down the nerve agents into less toxic components (47,69,70,100-104).
4.6. Centrally incapacitating agents. Centrally incapacitating agents are psychoactive drugs that modulate the functions of the central nervous system. They are meant to render individuals incapable of performing their duties without causing death. In the context of CWC-related debates, these chemicals are also referred to as Central Nervous System (CNS)-Acting Chemicals. A variety of chemicals with different mechanisms of action and different effects have been explored for their potential use as incapacitating agents, including, among others, psychedelic drugs (e.g. N,N-diethyl lysergamide, or LSD), stimulants, sedatives, narcotic analgesics, and anticholinergic drugs. Most of these chemicals are dual use substances that, beyond their use as incapacitating agents, have pharmaceutical applications for the treatment of mental conditions. All centrally incapacitating agents are chemicals endowed with the ability to readily cross the blood-brain barrier, a characteristic that is absolutely essential to allow their activity on the central nervous system. Moreover, to exert their activity on the central nervous system, centrally incapacitating agents must typically be administered systemically. Common ways of administration include injections, oral ingestion, or inhalation of aerosols. Unlike peripheral chemosensory irritants, centrally incapacitating agents tend to produce effects that last a rather long time, typically hours or days. For this reason, and because, although safe when used for therapeutic purposes under medical control, they can have very low safety margins when delivered as aerosols for incapacitating purposes (as it is difficult to predict the administered dose and, in some cases, the agents have extremely low LD50 values), the OPCW Scientific Advisory Board advised that centrally acting incapacitants cannot be regarded as meeting the CWC definition of riot control agents. Here we will discuss two centrally incapacitating agents, namely the anticholinergic drug BZ, and the opioid drug fentanyl, which exert their action by modulating the activity of two families of G protein-coupled receptors, namely the muscarinic acetylcholine and the opioid receptors, respectively (Fig. 9) (31,40,72,105).

Fig. 9. Examples of centrally incapacitating agents.
4.6.1 BZ and fentanyl. Anticholinergic drugs arechemicals that block the action of the neurotransmitter acetylcholine. In particular, 3-quinuclidinyl benzylate, also known as BZ, is a non-selective antagonist of the muscarinic acetylcholine receptors, just like atropine. BZ, however, is more potent. The first effects of BZ are on the peripheral nervous system and include decreased secretions, drowsiness, tachycardia, and mydriasis (pupil dilation). After a few hours, the central effects begin, the most severe of which include the development of hallucinations and erratic behavior and the interference with cognitive functions. Reversible inhibitors of acetylcholinesterase, such as physostigmine, can be used as antidotes, as they cause an increase of the synaptic concentration of acetylcholine, thus contrasting the anticholinergic action of BZ. Maintaining hydration and cooling are also important countermeasures to contrast the peripheral anticholinergic effects of BZ. Of note, BZ is a CWC Schedule 2 compound. Hence, its use as a riot control agent is explicitly prohibited by the CWC (31,40,72,105).
Fentanyl is an agonist of the opioid receptors, just like morphine, of which it is about 300 times more potent. It is a narcotic analgesic chemical, which also produces euphoria. Like nerve agents, it causes miosis. Importantly, due to its stimulation of the m subtypes of the opioid receptors, exposure to fentanyl can lead to central respiratory depression, with lethal consequences. This can be contrasted with the administration of opioid antagonists that act primarily at the m opioid receptors, such as naloxone, or agonists of the serotonin 5-HT4 receptors, which stimulate cells to produce cyclic adenosine monophosphate (cAMP), thus contrasting the inhibition of cAMP production caused by the m opioid receptors. The risks of using fentanyl as an incapacitating agent are highlighted by the tragic events of the 2002 Moscow theatre events. To resolve a situation in which about 800 people had been taken hostage by Chechen rebels inside a theatre, Russian security forces allegedly pumped into the theatre fentanyl vapors, causing the death of 119 of the hostages. Although there are still questions about the exact nature of the deployed substance or mixture of substances, chemical analysis on clothing identified a mixture of two fentanyl analogs, carfentanil and remifentanil. Moreover, norcarfentanil, a metabolite probably originating from carfentanil, was found in a urine sample (31,40,105,106).
General Readings
Hersman, R. K. C., and Pittinos, W. Restoring Restraint: Enforcing Accountability for Users of Chemical Weapons. Rowman and Littlefield, Lanham, 2018. Available online at:
https://www.csis.org/analysis/restoring-restraint
Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition. Lukey, B. J., Romano, J. A., and Salem, H. eds. CRC Press, 2019.
Medical Aspects of Chemical Warfare. Tuorinsky, S. D. ed. Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, 2008. Available online at:
https://www.cs.amedd.army.mil/Portlet.aspx?id=d3d11f5a-f2ef-4b4e-b75b-6ba4b64e4fb2
Practical Guide for Medical Management of Chemical Warfare Casualties. Organisation for the Prohibition of Chemical Weapons (OPCW), 2019. Available online at:
Cited References
1. Organisation for the Prohibition of Chemical Weapons (OPCW). The Convention on the Prohibition of the Development, Production, Stockpiling and Use of Chemical Weapons and on their Destruction (https://www.opcw.org/chemical-weapons-convention/).
2. Mumy, K. L., Howard, W. R., Parker, A., Forman, J., and Winfield, G. (2019) Chapter 4. Organization for the Prohibition of Chemical Weapons (OPCW): History, Mission, and Accomplishments. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 59-69
3. Hart, J., and Stock, T. (2019) Chapter 5. Chemical Weapons Holdings and Their Internationally Verified Destruction. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 71-85
4. Bunn, G. (1970) Gas and Germ Warfare: International Legal History and Present Status. Proc Natl Acad Sci U S A 65, 253-260
5. Nixon, R. M. (1969) Statement on Chemical and Biological Defense Policies and Programs. Public Papers of the Presidents of the United States
6. Balali-Mood, M., Mathews, R., Pita, R., Rice, P., Romano, J. A., Thiermann, H., and Willems, J. L. (2019) Chapter 1: Introduction and Historical Overview. in Practical Guide for Medical Management of Chemical Warfare Casualties, Organisation for the Prohibition of Chemical Weapons (OPCW) (https://www.opcw.org/sites/default/files/documents/2019/05/Full%20version%202019_Medical%20Guide_WEB.pdf). pp 9-21
7. United Nations Office for Disarmament Affairs (UNODA). Convention on the Prohibition of the Development, Production and Stockpiling of Bacteriological (Biological) and Toxin Weapons and on Their Destruction (http://disarmament.un.org/treaties/t/bwc/text).
8. Hersman, R. K. C., and Pittinos, W. (2018) Restoring Restraint: Enforcing Accountability for Users of Chemical Weapons, Rowman and Littlefield, Lanham
9. International Partnership Against Impunity for the Use of Chemical Weapons. (https://www.noimpunitychemicalweapons.org/-en-.html).
10. Salem, H., Ternay, A. L., and Smart, J. K. (2019) Chapter 1. Brief History and Use of Chemical Warfare Agents in Warfare and Terrorism. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 3-16
11. Eisenkraft, A., and Falk, A. (2019) Chapter 7. The Use of Chemical Warfare Agents during the Syrian Civil War. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 103-122
12. Jones, M. R. (2019) Chapter 17. Personal Protective Equipment (PPE): Practical and Theoretical Considerations. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 303-372
13. Capacio, B. R., Smith, J. R., Gordon, R. K., Haigh, J. R., Barr, J. R., and Platoff, G. E. (2008) Chapter 22: Medical Diagnostics. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 691-751
14. (2019) Section IV. Detection. in Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 399-546
15. (2019) Section V. Decontamination. in Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 547-574
16. Williams, P., Willens, S., Anderson, J., Adler, M., and Hilmas, C. J. (2008) Chapter 19: Toxins: Established and Emergent Threats. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 613-644
17. Henderson, T. J., Emmett, G., Dorsey, R. M., Millard, C. B., LeClaire, R. D., and Salem, H. (2019) Chapter 13. The Structural Biology, Biochemistry, Toxicology, and Military Use of the Ricin Toxin and the Associated Treatments and Medical Countermeasures for Ricin Exposure. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 203-226
18. Adler, M., Singh, A. K., Gul, N., and Lebeda, F. J. (2019) Chapter 14. Medical Countermeasures for Intoxication by Botulinum Neurotoxin. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 227-244
19. Balali-Mood, M., Mathews, R., Pita, R., Rice, P., Romano, J. A., Thiermann, H., and Willems, J. L. (2019) Chapter 2: General Considerations in Management of Chemical Casualties. in Practical Guide for Medical Management of Chemical Warfare Casualties, Organisation for the Prohibition of Chemical Weapons (OPCW) (https://www.opcw.org/sites/default/files/documents/2019/05/Full%20version%202019_Medical%20Guide_WEB.pdf). pp 23-40
20. Balali-Mood, M., Mathews, R., Pita, R., Rice, P., Romano, J. A., Thiermann, H., and Willems, J. L. (2019) Chapter 8: Toxic Chemicals of Biological Origin. in Practical Guide for Medical Management of Chemical Warfare Casualties, Organisation for the Prohibition of Chemical Weapons (OPCW) (https://www.opcw.org/sites/default/files/documents/2019/05/Full%20version%202019_Medical%20Guide_WEB.pdf). pp 107-122
21. Hayes, A. W., and Dixon, D. (2017) Cornerstones of Toxicology. Toxicol Pathol 45, 57-63
22. Organisation for the Prohibition of Chemical Weapons (OPCW). Chemical Weapons Convention – Annex on Chemicals (https://www.opcw.org/chemical-weapons-convention/annexes/annex-chemicals/annex-chemicals).
23. Organisation for the Prohibition of Chemical Weapons (OPCW). Chemical Weapons Convention – Verification Annex, Part VI (https://www.opcw.org/chemical-weapons-convention/annexes/verification-annex/part-vi-regime-schedule-1-chemicals-and).
24. Organisation for the Prohibition of Chemical Weapons (OPCW). Chemical Weapons Convention – Verification Annex, Part VII (https://www.opcw.org/chemical-weapons-convention/annexes/verification-annex/part-vii-regime-schedule-2-chemicals-and).
25. Organisation for the Prohibition of Chemical Weapons (OPCW). Chemical Weapons Convention – Verification Annex, Part VIII (https://www.opcw.org/chemical-weapons-convention/annexes/verification-annex/part-viii-regime-schedule-3-chemicals-and).
26. Pitschmann, V. (2014) Overall view of chemical and biochemical weapons. Toxins 6, 1761-1784
27. Mauroni, A., and Blades, T. A. (2019) Chapter 6. Syria’s Chemical Disposal Program. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 87-102
28. Tucker, J. B. (2007) Chapter 15. Peace and War. in War of nerves, Anchor. pp
29. Olson, K. B. (1999) Aum Shinrikyo: Once and Future Threat? Emerging Infectious Diseases 5, 513-516
30. Hilmas, C. J., Smart, J. K., and Hill, B. A. (2008) Chapter 2: History of Chemical Warfare. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 9-76
31. Timperley, C. M., Forman, J. E., Aas, P., Abdollahi, M., Benachour, D., Al-Amri, A. S., Baulig, A., Becker-Arnold, R., Borrett, V., Carino, F. A., Curty, C., Gonzalez, D., Geist, M., Kane, W., Kovarik, Z., Martinez-Alvarez, R., Mikulak, R., Mourao, N. M. F., Neffe, S., Nogueira, E. D., Ramasami, P., Raza, S. K., Rubaylo, V., Saeed, A. E. M., Takeuchi, K., Tang, C., Trifiro, F., van Straten, F. M., Suarez, A. G., Waqar, F., Vanninen, P. S., Zafar-Uz-Zaman, M., Vucinic, S., Zaitsev, V., Zina, M. S., Holen, S., and Izzati, F. N. (2018) Advice from the Scientific Advisory Board of the Organisation for the Prohibition of Chemical Weapons on Riot Control Agents in Connection to the Chemical Weapons Convention. Rsc Adv 8, 41731-41739
32. International Committee of the Red Cross (ICRC) Customary IHL Database. Rule 76. Herbicides (https://ihl-databases.icrc.org/customary-ihl/eng/docs/v1_rul_rule76).
33. International Committee of the Red Cross (ICRC) Customary IHL Database. Practice Relating to Rule 76. Herbicides (https://ihl-databases.icrc.org/customary-ihl/eng/docs/v2_rul_rule76).
34. Pandit, N. K. (2006) Chapter 14: Plasma Concentration-Time Curves. in Introduction to the Pharmaceutical Sciences, Lippincott Williams and Wilkins, Lanham. pp 213-224
35. Rozman, K. K. (2019) Approaches for Using Toxicokinetic Information in Assessing Risk to Deployed U.S. Forces. in Strategies to Protect the Health of Deployed U.S. Forces. Assessing Health Risks to Deployed U.S. Forces. (Lukey, B. J., Romano, J. A., and Salem, H. eds.), National Academi Press, Washington, D.C. pp 113-149
36. Pandit, N. K. (2006) Chapter 17: Dose-Response Ralationships. in Introduction to the Pharmaceutical Sciences, Lippincott Williams and Wilkins, Lanham. pp 277-290
37. Hulet, S. W., Dabisch, P. A., Kristovich, R. L., Sommerville, D. R., and Mioduszewski, R. J. (2019) Chapter 11: Inhalation Toxicology of Chemical Agents. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 163-180
38. Tenberge, W. F., Zwart, A., and Appelman, L. M. (1986) Concentration-Time Mortality Response Relationship of Irritant and Systemically Acting Vapors and Gases. Journal of Hazardous Materials 13, 301-309
39. Sweeney, L. M., Sommerville, D. R., Channel, S. R., Sharits, B. C., Gargas, N. M., and Gut, C. P., Jr. (2015) Evaluating the Validity and Applicable Domain of the Toxic Load Model: Impact of Concentration vs. Time Profile on Inhalation Lethality of Hydrogen Cyanide. Regul Toxicol Pharmacol 71, 571-584
40. Katz, S. A., and Salem, H. (2019) Chapter 15: Incapacitating Agents and Technologies: A Review. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 246-276
41. Loftsson, T. (2015) Chapter 3: Physicochemical Properties and Pharmacokinetics. in Essential Pharmacokinetics. A Primer for Pharmaceutical Scientists, Academic Press. pp 85-104
42. Tuorinsky, S. D., and Sciuto, A. M. (2008) Chapter 10: Toxic Inhalational Injury and Toxic Industrial Chemicals. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 339-370
43. Wagner, G. W., and MacIver, B. K. (1998) Degradation and fate of mustard in soil as determined by C-13 MAS NMR. Langmuir 14, 6930-6934
44. Rockwood, G. A., Platoff, G. E., and Salem, H. (2019) Chapter 12: Cyanides: Toxicology, Clinical Presentation, and Medical Management in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 182-202
45. Journal of the Royal Army Med Corps. (2002) Chemical casualties. Lung damaging agents (choking agents). 148, 377-382
46. Pardridge, W. M. (2012) Drug Transport Across the Blood-Brain Barrier. J Cereb Blood Flow Metab 32, 1959-1972
47. Kassa, J. (2019) Chapter 32. Oxime Research. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 681-696
48. Hurst, C. G., Petrali, J. P., Barillo, D. J., Graham, J. S., Smith, W. J., Urbanetti, J. S., and Sidell, F. R. (2008) Chapter 8: Vesicants. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 259-309
49. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Bis(2-chloroethyl)sulfide (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+336).
50. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Mechlorethamine (HN2) (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+5083).
51. Toxic Substances Portal – Agency for Toxic Substances and Disease Registry. Nitrogen Mustard (HN1, HN2, HN3). https://www.atsdr.cdc.gov/mmg/mmg.asp?id=920&tid=189.
52. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Lewisite (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+6393).
53. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Chlorine (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+206).
54. Sommerville, D. R., Bray, J. J., Reutter-Christy, S. A., Jablonski, R. E., and Shelly, E. E. (2009) Review and Assessment of Chlorine Mammalian Lethality Data and the Development of a Human Estimate – R1. Department Of Homeland Security, Chemical Security Analysis Center
55. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Phosgene (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+796).
56. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Perfluoroisobutylene (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+7708).
57. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Chloropicrin (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+977).
58. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. 2-Chloroacetophenone (CN). (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+972).
59. Hilmas, C. J., Poole, M. J., Katos, A. M., and Williams, P. T. (2009) Chapter 12: Riot Control Agents. in Handbook of Toxicology of Chemical Warfare Agents (Gupta, R. C. ed.), Academic Press. pp 153-175
60. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. 2-Chlorobenzalmalononitrile (CS) (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+4346).
61. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Dibenz(b,f)[1,4]oxazepine (CR) (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+7598).
62. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Capsaicin (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+954).
63. Baskin, S. I., Kelly, J. B., Maliner, B. I., Rockwood, G. A., and Zoltani, C. K. (2008) Chapter 11: Cyanide Poisoning. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 371-410
64. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Hydrogen Cyanide (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+165).
65. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Cyanogen Chloride (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+917).
66. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Arsine (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+510).
67. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Hydrogen Sulfide (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+576).
68. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Carbon Monoxide (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+903).
69. Costanzi, S., Machado, J. H., and Mitchell, M. (2018) Nerve Agents: What They Are, How They Work, How to Counter Them. ACS chemical neuroscience 9, 873-885
70. Sidell, F. R., Newmark, J., and McDonough, J. H. (2008) Chapter 5: Nerve Agents. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 155-219
71. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. BZ (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+7533).
72. Ball, J. C. (2015) Dual use research of concern: derivatives of 3-quinuclidinyl benzilate (BZ). Mil. Med. Sci. Lett. 84, 2-41
73. Committee on Acute Exposure Guideline Levels; Committee on Toxicology; Board on Environmental Studies and Toxicology; Division on Earth and Life Studies; National Research Council. (2013) Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 14. Appendix I. Agent BZ (3-Quinuclidinyl Benzilate): Acute Exposure Guideline Levels (https://www.nap.edu/catalog/18313/acute-exposure-guideline-levels-for-selected-airborne-chemicals-volume-14). The National Academies Press.
74. Hazardous Substances Data Bank (HSDB), National Library of Medicine’s TOXNET system. Fentanyl (http://toxnet.nlm.nih.gov/cgi-bin/sis/search2/r?dbs+hsdb:@term+@DOCNO+3329).
75. Lichtman, A. H. (1998) The Up-and-Down Method Substantially Reduces the Number of Animals Required to Determine Antinociceptive ED50 Values. Journal of pharmacological and toxicological methods 40, 81-85
76. Manral, L., Muniappan, N., Gupta, P. K., Ganesan, K., Malhotra, R. C., and Vijayaraghavan, R. (2009) Effect of Exposure to Fentanyl Aerosol in Mice on Breathing Pattern and Respiratory Variables. Drug Chem Toxicol 32, 108-113
77. Ciottone, G. R. (2018) Toxidrome Recognition in Chemical-Weapons Attacks. N Engl J Med 378, 1611-1620
78. Pulley, S. A. (2019) Chapter 30: Medical Management of Chemical Warfare Agents. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 627-654
79. Malaviya, R., Heck, D. E., Casillas, R. P., Laskin, J. D., and Laskin, D. L. (2019) Mustard Vesicants. in Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 131-144
80. Balali-Mood, M., Mathews, R., Pita, R., Rice, P., Romano, J. A., Thiermann, H., and Willems, J. L. (2019) Chapter 3: Vesicants (Blister Agents). in Practical Guide for Medical Management of Chemical Warfare Casualties, Organisation for the Prohibition of Chemical Weapons (OPCW) (https://www.opcw.org/sites/default/files/documents/2019/05/Full%20version%202019_Medical%20Guide_WEB.pdf). pp 41-58
81. Spain, P. D., and Kadan-Lottick, N. (2012) Observations of Unprecedented Remissions Following Novel Treatment for Acute Leukemia in Children in 1948. J R Soc Med 105, 177-181
82. Shakarjian, M. P., Heck, D. E., Gray, J. P., Sinko, P. J., Gordon, M. K., Casillas, R. P., Heindel, N. D., Gerecke, D. R., Laskin, D. L., and Laskin, J. D. (2010) Mechanisms Mediating the Vesicant Actions of Sulfur Mustard after Cutaneous Exposure. Toxicol Sci 114, 5-19
83. Siegert, M., Gandor, F., Kranawetvogl, A., Borner, H., Thiermann, H., and John, H. (2019) Methionine(329) in Human Serum Albumin: A Novel Target for Alkylation by Sulfur Mustard. Drug Test Anal 11, 659-668
84. Hurst, C. G., and Smith, W. J. (2010) Health Effects of Exposure to Vesicant Agents. in Chemical Warfare Agents: Chemistry, Pharmacology, Toxicology, and Therapeutics – Second Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), Informa Healthcare. pp 293-313
85. Sezigen, S., Ivelik, K., Ortatatli, M., Almacioglu, M., Demirkasimoglu, M., Eyison, R. K., Kunak, Z. I., and Kenar, L. (2019) Victims of Chemical Terrorism, a Family of Four who Were Exposed to Sulfur Mustard. Toxicol Lett 303, 9-15
86. Balali-Mood, M., Mathews, R., Pita, R., Rice, P., Romano, J. A., Thiermann, H., and Willems, J. L. (2019) Chapter 5: Lung-Damaging (Choking) Agents. in Practical Guide for Medical Management of Chemical Warfare Casualties, Organisation for the Prohibition of Chemical Weapons (OPCW) (https://www.opcw.org/sites/default/files/documents/2019/05/Full%20version%202019_Medical%20Guide_WEB.pdf). pp 73-84
87. Salem, H., Gutting, B. W., Kluchinsky, T. A., Boardman, C. H., Tuorinsky, S. D., and Hout, J. J. (2008) Chapter 13: Riot Control Agents. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 441-484
88. Smith, M. G., Stone, W., Guo, R.-F., Ward, P. A., Suntres, Z., Mukherjee, S., and Das, S. K. (2010) Chapter 12: Vesicants and Oxidative Stress. in Chemical warfare agents: chemistry, pharmacology, toxicology, and therapeutics (Lukey, B. J., Romano, J. A., and Salem, H. eds.), Informa Healthcare. pp 248-292
89. Balali-Mood, M., Mathews, R., Pita, R., Rice, P., Romano, J. A., Thiermann, H., and Willems, J. L. (2019) Chapter 7: Riot Control Agents. in Practical Guide for Medical Management of Chemical Warfare Casualties, Organisation for the Prohibition of Chemical Weapons (OPCW) (https://www.opcw.org/sites/default/files/documents/2019/05/Full%20version%202019_Medical%20Guide_WEB.pdf). pp 99-106
90. Venkatachalam, K., and Montell, C. (2007) TRP Channels. Annu Rev Biochem 76, 387-417
91. Balali-Mood, M., Mathews, R., Pita, R., Rice, P., Romano, J. A., Thiermann, H., and Willems, J. L. (2019) Chapter 6: Blood Agents (Cyanide Compounds). in Practical Guide for Medical Management of Chemical Warfare Casualties, Organisation for the Prohibition of Chemical Weapons (OPCW) (https://www.opcw.org/sites/default/files/documents/2019/05/Full%20version%202019_Medical%20Guide_WEB.pdf). pp 85-97
92. Antonini, E., Brunori, M., Greenwood, C., Malmstrom, B. G., and Rotilio, G. C. (1971) The Interaction of Cyanide with Cytochrome Oxidase. Eur J Biochem 23, 396-400
93. Balali-Mood, M., Mathews, R., Pita, R., Rice, P., Romano, J. A., Thiermann, H., and Willems, J. L. (2019) Chapter 4: Nerve Agents. in Practical Guide for Medical Management of Chemical Warfare Casualties, Organisation for the Prohibition of Chemical Weapons (OPCW) (https://www.opcw.org/sites/default/files/documents/2019/05/Full%20version%202019_Medical%20Guide_WEB.pdf). pp 59-72
94. Leikin, J. B., Thomas, R. G., Walter, F. G., Klein, R., and Meislin, H. W. (2002) A Review of Nerve Agent Exposure for the Critical Care Physician. Crit Care Med 30, 2346-2354
95. Chemical Hazards Emergency Medical Management (CHEMM), National Library of Medicine,. (2019) Fourth Generation Agents – Medical Management Guidelines. https://chemm.nlm.nih.gov/nerveagents/FGA_Medical_Management_Guidelines_508.pdf.
96. Costanzi, S., and Koblentz, G. (2019) Controlling Novichok after Salisbury: revising the Chemical Weapons Convention schedules. The Nonproliferation Review, doi: 10.1080/10736700.10732019.11662618. https://doi.org/10736710.10731080/10736700.10732019.11662618
97. Organisation for the Prohibition of Chemical Weapons (OPCW). Recommendation for a Change to Schedule 1 of the Annex on Chemicals to the Chemical Weapons Convention, EC-M-62/DEC.1, 14 January 2019 (https://www.opcw.org/sites/default/files/documents/2019/01/ecm62dec01%2B%28e%29.pdf).
98. Adler, M., Apland, J. P., Deshpande, S. S., Koplovitz, I., and Lenz, D. E. (2019) Chapter 18. Pyridostigmine Protection from Nerve Agent Intoxication: Concept to FDA Approval. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 373-392
99. Timperley, C. M., Forman, J. E., Abdollahi, M., Al-Amri, A. S., Baulig, A., Benachour, D., Borrett, V., Carino, F. A., Geist, M., Gonzalez, D., Kane, W., Kovarik, Z., Martinez-Alvarez, R., Mourao, N. M. F., Neffe, S., Raza, S. K., Rubaylo, V., Suarez, A. G., Takeuchi, K., Tang, C., Trifiro, F., van Straten, F. M., Vanninen, P. S., Vucinic, S., Zaitsev, V., Zafar-Uz-Zaman, M., Zina, M. S., and Holen, S. (2019) Advice on Assistance and Protection Provided by the Scientific Advisory Board of the Organisation for the Prohibition of Chemical Weapons: Part 1. On Medical Care and Treatment of Injuries from Nerve Agents. Toxicology 415, 56-69
100. Goldsmith, M., Ashani, Y., Otto, T. C., Cadieux, C. L., and Riddle, D. S. (2019) Chapter 31. Protein-Based Bioscavengers of Organophosphorus Nerve Agents. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 655-680
101. Chambers, J. E., and Meek, E. C. (2019) Chapter 33. Brain-Penetrating Reactivators of Organophosphate-Inhibited Acetylcholinesterase. in Chemical Warfare Agents. Biomedical and Psychological Effects, Medical Countermeasures, and Emergency Response – Third Edition (Lukey, B. J., Romano, J. A., and Salem, H. eds.), CRC Press. pp 697-702
102. Albuquerque, E. X., Pereira, E. F., Aracava, Y., Fawcett, W. P., Oliveira, M., Randall, W. R., Hamilton, T. A., Kan, R. K., Romano, J. A., Jr., and Adler, M. (2006) Effective Countermeasure Against Poisoning by Organophosphorus Insecticides and Nerve Agents. Proc Natl Acad Sci U S A 103, 13220-13225
103. Timperley, C. M., Abdollahi, M., Al-Amri, A. S., Baulig, A., Benachour, D., Borrett, V., Carino, F. A., Geist, M., Gonzalez, D., Kane, W., Kovarik, Z., Martinez-Alvarez, R., Fusaro Mourao, N. M., Neffe, S., Raza, S. K., Rubaylo, V., Suarez, A. G., Takeuchi, K., Tang, C., Trifiro, F., van Straten, F. M., Vanninen, P. S., Vucinic, S., Zaitsev, V., Zafar-Uz-Zaman, M., Zina, M. S., Holen, S., Forman, J. E., Alwan, W. S., and Suri, V. (2019) Advice on Assistance and Protection from the Scientific Advisory Board of the Organisation for the Prohibition of Chemical Weapons: Part 2. On Preventing and Treating Health Effects from Acute, Prolonged, and Repeated Nerve Agent Exposure, and the Identification of Medical Countermeasures Able to Reduce or Eliminate the Longer Term Health Effects of Nerve Agents. Toxicology 413, 13-23
104. Ross, M. C., Broomfield, C. A., Cerasoli, D. M., Doctor, B. P., Lenz, D. E., Maxwell, D. M., and Saxena, A. (2008) Chapter 7: Nerve Agent Bioscavenger: Development of a New Approach to Protect Against Organophosphorus Exposure. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 243-257
105. Ketchum, J. S., and Salem, H. (2008) Chapter 12: Incapacitating Agents. in Medical Aspects of Chemical Warfare (Tuorinsky, S. D. ed.), Office of The Surgeon General United States Army and Borden Institute, Walter Reed Army Medical Center, Falls Church, Virginia and Washington, D.C. pp 411-439
106. Riches, J. R., Read, R. W., Black, R. M., Cooper, N. J., and Timperley, C. M. (2012) Analysis of Clothing and Urine from Moscow Theatre Siege Casualties Reveals Carfentanil and Remifentanil Use. J Anal Toxicol 36, 647-656
